
Contact
Cancer is caused by defects in the normal genetic code. Emerging evidence suggests that regulatory mishaps during DNA replication are often the causative event for this dreadful disease. To address the fundamental relationship between DNA replication and cancer, my laboratory studies the major regulatory hub of DNA replication – the Mcm2-7 replicative helicase. As a helicase, Mcm2-7 uses ATP binding and hydrolysis to unwind double-stranded DNA to generate templates for DNA polymerase. From a regulatory standpoint, the loading, activation, and subsequent DNA unwinding by this complex are all heavily regulated to ensure the precise production of a single exact copy of the genome during each S-phase. Alterations in this regulation cause damage that has been shown experimentally to lead to cancer. In combination with various collaborators, my lab uses a multitude of biochemical, genetic, structural, cytological and genomic approaches to address fundamental biological issues using the budding yeast S. cerevisiae, an easy-to-work-with model eukaryote whose DNA replication process is conserved with those of humans. The fusion of complementary in vitro and in vivo methodologies in my lab provides excellent and unique research training opportunities. 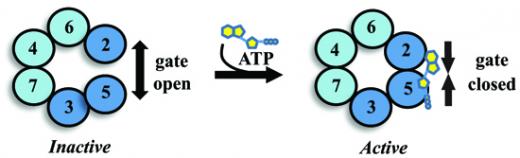
The key to Mcm2-7 regulation is its unusual subunit composition. Unlike related helicases formed from six identical subunits, Mcm2-7 is formed from six unique and functionally distinct subunits. Three Mcm subunits are specialized to unwind DNA (Mcm4, 6, and 7) while the other three (Mcm2, 3, and 5) regulate the activity of the complex. How the regulatory subunits control Mcm2-7 activity is a critical unsolved question in the replication field. To address this problem, we have found that the regulatory subunits form a reversible discontinuity within the Mcm2-7 ring structure (Figure 1). Biochemical activity is inversely related to the discontinuity – closing the ring activates DNA unwinding; while opening the ring inactivates this activity. These observations suggest that Mcm2-7 regulation in living cells might occur through cellular factors that regulate the state of the discontinuity. Recent structural data from our colleagues provide strong support for our hypothesis (Costa et. al. 2011 Nat. Struct. Biol. 18:471).
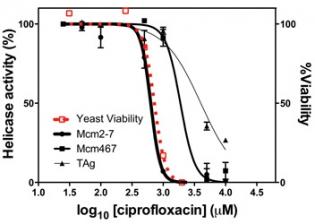
Although this model provides a rational mechanistic explanation for Mcm2-7 regulation, its relevance in living cells remains to be tested. Current studies in the Schwacha Lab largely focus on this issue by comparing various aspects of Mcm2-7 regulation within living cells with their biochemical effect on the Mcm2-7 discontinuity. In parallel, we have started to identify small molecule inhibitors of Mcm2-7 DNA unwinding activity (Figure 2). These compounds are ultimately hoped to provide both useful research tools to study Mcm function as well as therapeutic agents to fight cancer.
For a more complete discussion of current research in the Schwacha Lab, please visit our lab webpage
- Sriram Vijayraghavan, Past Grad
- Jordan Sanders, Graduate Student
- Nick Wechter, Research Assistant
