
Contact
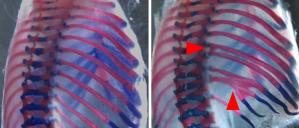
Birth defects result from errors made during embryonic development. Normal development involves careful orchestration of multiple events, including changes in gene transcription, cell shape, proliferation, and tissue morphogenesis. Alterations in the developmental program can have dramatic effects on the organism. Sometimes these effects are so drastic that lethality occurs early in the development. Less severe errors may allow the organism to develop, but lead to devastating birth defects. We are interested in how different mesoderm populations arise during development and thus we focus on how mesoderm, produced in the primitive streak, acquires different cell fates and behaviors. We are particularly interested in the specification and development of paraxial mesoderm, which forms somites and ultimately the ribs, vertebrae and skeletal muscle. Improper formation and patterning of paraxial mesoderm can lead to birth defects such as scoliosis, fusions of the ribs (Figure 1) and improper muscles segmentation (Figure 2).
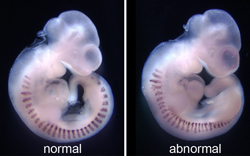
During embryogenesis, the primitive streak (PS) generates the paraxial mesoderm (PAM), which undergoes segmentation to form the somites and ultimately the vertebrae, ribs, skeletal muscle and dermis. We are studying the roles that two T-box transcription factors, T and Tbx6, play in mesoderm formation and cell fate decisions. T-box proteins share a similar DNA binding domain, the T-domain, and therefore can bind similar sequences in vitro. T and Tbx6 are both expressed in the PS, but also have unique areas of expression: T in the notochord and Tbx6 in the presomitic PAM. We hypothesize that T is necessary to maintain cells in a PS-like state and enables cells to transition from an epithelial to a mesenchymal cell morphology. T is necessary for continued production of mesoderm and extension of the body axis, while Tbx6 is required to push cells toward a PAM fate.

We use the mouse as a model system to understand how PAM is generated from the PS. The mouse offers a number of advantages, including the ability to routinely inactivate genes and to create transgenic animals that over- or misexpress genes of interest. Because of the close relationship between the mouse and human, it is possible to directly apply things we learn in the mouse to human development. Genetic studies reveal that maintaining the proper levels of T and Tbx6 is critical for normal development and that these factors can compete for binding to downstream targets. We hypothesize that altering the relative levels of T and Tbx6 allows opportunistic binding to the other’s targets and this contributes to the observed phenotypes. We are testing this hypothesis using transgenic approaches to misexpress T and Tbx6 in the embryo. We also use molecular approaches, including luciferase transcriptional assays to examine how T and Tbx6 function alone and together to affect target gene expression and chromatin immunoprecipitations to examine in vivo binding of these factors to target gene enhancers. These studies will unravel how T and Tbx6 direct cell fate by identifying their target genes and finally testing how target gene expression is altered in T and Tbx6 transgenic embryos.
- Mariah Denhart, Graduate Student
- Sonya Acharya, Undergraduate Researcher
- Aloysius Snuffleupagus, Deb's Imaginary Friend
